|
They have a positive influence on endoplasmic reticulum (ER) stress-related pathologies. They are termed as chemical chaperons that help to treat several metabolic disorders, such as obesity, type 2 diabetes, and atherosclerosis. These molecules inhibit the signaling pathways involved in the growth and proliferation of tumor cells. Hsp 90 is a type of chaperon which has various therapeutic importance. Both Hsp 70 and 60 have two different forms, where the first form is the binding form and the second form is the folding-active state. They have 14 different components with two rings, made up of seven rings. This quarantine mechanism helps to prevent the proteins from getting clumped with other linear polypeptide chains in the cytoplasm. Hsp 60, unlike Hsp 70, which is involved in the prevention of protein aggregation, their function is to isolate unfolded proteins. These Hsp70 proteins recognize the “extended region” with the hydrophobic region of the unfolded protein, and prevent the random aggregation. The hydrolysis of ATP occurring at the N terminal of the chaperon molecule helps in the opening and binding of the substrate to the C terminal. They are monomers with a C and an N domain the C terminal domain binds with the substrate protein, while the N terminal domain contains ATPase. Hsp70 acts as a catalyst in protein folding processes, such as misfolding and refolding of the aggregated proteins, assembling and folding of newly synthesized proteins. The name heat shock protein was first identified from a bacterium that is at high stressful states such as high pH, temperature, and hypoxic conditions. The exposure of the heat destabilizes the protein structure, so during such harsh conditions they need assistance to fold. 37, 1202–1208 (2016).Chaperone molecules are termed as "heat shock" proteins (HSPs), as they are formed in a large amount when the cells get exposed to heat. 5 characterize TDP-43 condensates formed in the cytoplasm under proteotoxic conditions and identify factors that may act as chaperones by inhibiting protein aggregation. In the current issue of Nature Cell Biology, Lu et al. Therefore, understanding the biogenesis of TDP-43 aggregates and how their assembly may be controlled is of major therapeutic interest. This pathology also accumulates in all cases of an Alzheimer’s-disease-associated dementia affecting the oldest population, known as limbic-predominant age-related TDP-43 encephalopathy (LATE) 4. In particular, neuronal and glial TDP-43 inclusions are found in nearly all individuals with amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) with ubiquitin-positive inclusions (FTLD-U) 3. The self-assembly of the RBP TDP-43 (TAR DNA-binding protein) in the cytoplasm has come under special scrutiny because TDP-43 aggregates in this compartment, and the corresponding nuclear depletion of the protein, are common features of various neurodegenerative disorders 3. 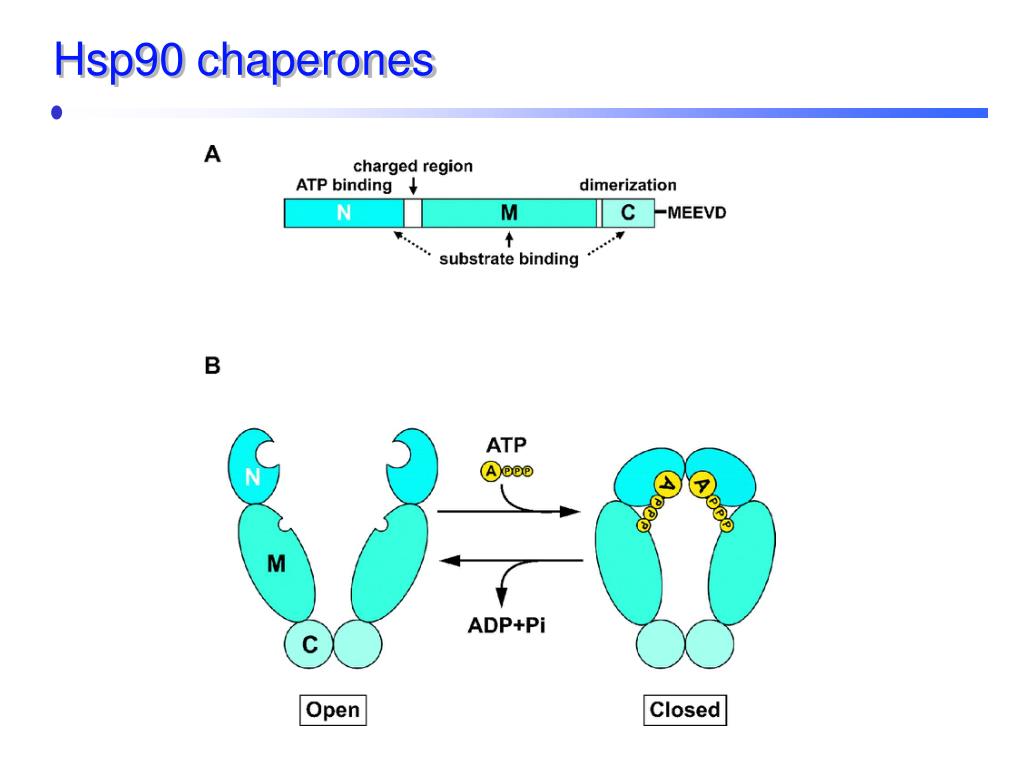 
Disruptions in the material properties of RBP condensates may generate irreversible aggregates linked to neurodegeneration 2. 
RNA-binding proteins (RBPs) are characteristically prone to phase separation, and their function is modulated by LLPS 1. Mounting evidence suggests that biomolecular condensation has a central role in numerous cellular functions and mechanisms of disease, including cancer and neurodegeneration. Biomolecular condensation is mediated by phase separation, or liquid–liquid phase separation (LLPS), which consists of the de-mixing of biological macromolecules such as proteins and nucleic acids into dense and light phases.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
